|
Preretinal hemorrhages are “boat” or ‘D’ shaped hemorrhages which collect between the posterior limiting membrane of the vitreous and internal limiting membrane (ILM) of the retina. They are commonly seen in choroidal neovascular membranes (CNVM), choroidal tumors, and choroidal rupture secondary to acute trauma. These hemorrhages are located between RPE and Bruch membrane and appear dark red with well-defined sharp borders. Sub-macular hemorrhages are commonly seen in choroidal neovascular membranes secondary to ARMD. Such hemorrhages are commonly seen in ARMD, presumed ocular histoplasmosis, high myopia, PCV, retinal macroaneurysm, and trauma. The hemorrhages are deep red in color and broader in shape with diffuse margins. 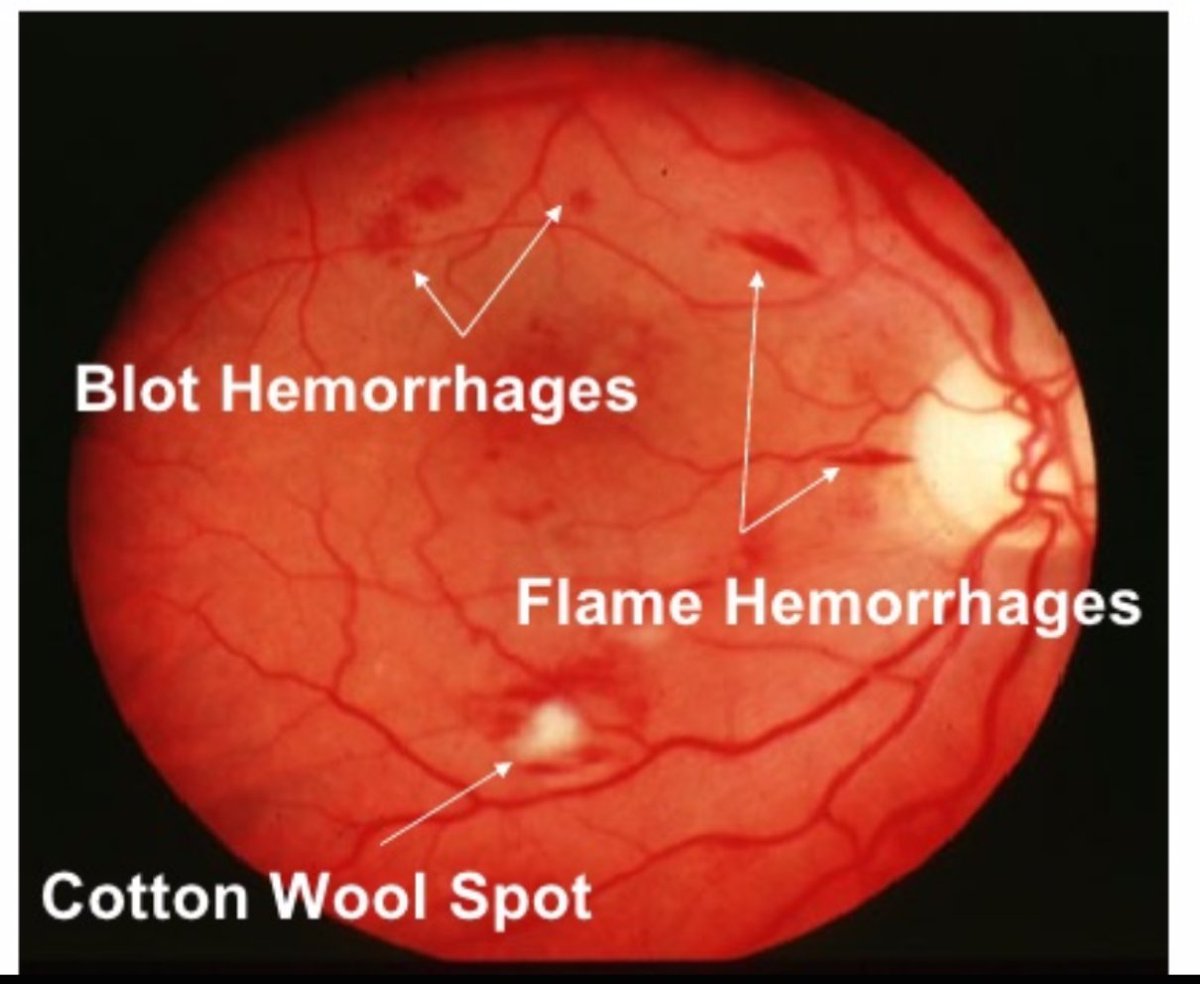
These hemorrhages occur between the photoreceptor layer and retinal pigment epithelium (RPE). Purtscher-like retinopathy is seen in acute pancreatitis, renal failure, and autoimmune disease. Retinal findings are intraretinal whitening, cotton wool spots, and intraretinal hemorrhages. Purtsher retinopathy is an occlusive microvasculopathy associated with cranial or thoracic compressive trauma. Common causes for such hemorrhages include diabetic retinopathy, retinal vein occlusions, ocular ischemic syndrome, sickle cell retinopathy, and juxta foveal telangiectasia. These are dense, dark red, sharply outlined, and are seen in disorders that affect the pre-venular deep capillary layer. These dot and blot hemorrhages are found within the inner nuclear and outer plexiform layers of the retina. Roth spots are characteristic of subacute bacterial endocarditis and also seen in leukemia, anemia, anoxia, and other rare conditions. Capillary rupture with extravasation, and central fibrin–platelet plug gives the white center to the Roth spot. It was Litten who described the association (Litten sign) and referred to it as Roth spots. Hypertension correlates with the severity of the diabetic retinopathy.Roth spots: Roth spots are round in shape with a white center. Monkeys with type 2 diabetes have many of the angiopathic changes associated with human diabetic retinopathy. This is the first detailed analysis of retinopathy in a colony of spontaneous type 2 diabetic monkeys. In elastase-digested retinas, the ratio of pericytes to endothelial cells was 0.66:1 in diabetic and 0.64:1 in nondiabetic (P = 0.75) retinas. There was a significant correlation between the occurrence of retinopathy and hypertension (P = 0.037 for systolic pressure P = 0.019 for diastolic pressure). 
There were apparent fluid-filled spaces in the outer plexiform layer in three of these maculas, suggesting macular edema. Formation of small intraretinal microvascular abnormalities (IRMAs) and microaneurysms were associated with the areas of nonperfusion. 
Large nonperfused areas extending from optic disc to midfovea were observed in four diabetic monkeys. Dot/blot hemorrhages, cotton-wool spots, and small nonperfused areas were the earliest histologically documented changes in the retinas. Tissue sections were cut through areas of interest.Ĭotton-wool spots, intraretinal hemorrhages, and hard exudates in the macula were observed by ophthalmoscopy in some diabetic monkeys. Retinas from 16 diabetic monkeys and 6 nondiabetic monkeys were incubated postmortem for adenosine diphosphatase (ADPase) activity (labels viable retinal blood vessels) and flat-embedded in JB-4. Ophthalmoscopic examinations were performed on aged normal and diabetic monkeys. The purpose of this study was to investigate clinically and histopathologically the ocular changes in these monkeys. Type 2 diabetes occurs spontaneously in rhesus monkeys and shows an extraordinary similarity to human diabetes in clinical features and relative time course.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
